In the field of chemistry, understanding the role of acidic hydrogen is crucial to studying bases, acids and chemical Reactivity. Acidic hydrogen is an atom of hydrogen in molecules that are quickly transferred to form proton (H+) in an chemical reaction. It is vital to identify acidic hydrogen to organic and biochemical chemistry and analytical chemistry as it can help predict the behavior of a reaction in terms of acidity strength, pH, as well as molecular stabilities. This article will help you determine acidic hydrogen, the types and the practical techniques used in labs.
1. Definition of Acidic Hydrogen
- Acidic hydrogen atom is linked to an atom that is electronegative or a part of the Polar bond.
- As the hydrogen ionsizes and forms proton (H+) and leaves behind the positively charged type (anion or conjugate base).
- Acidic hydrogens can be found in carboxylic acid, alcohols, phenols, and a few organic compounds.
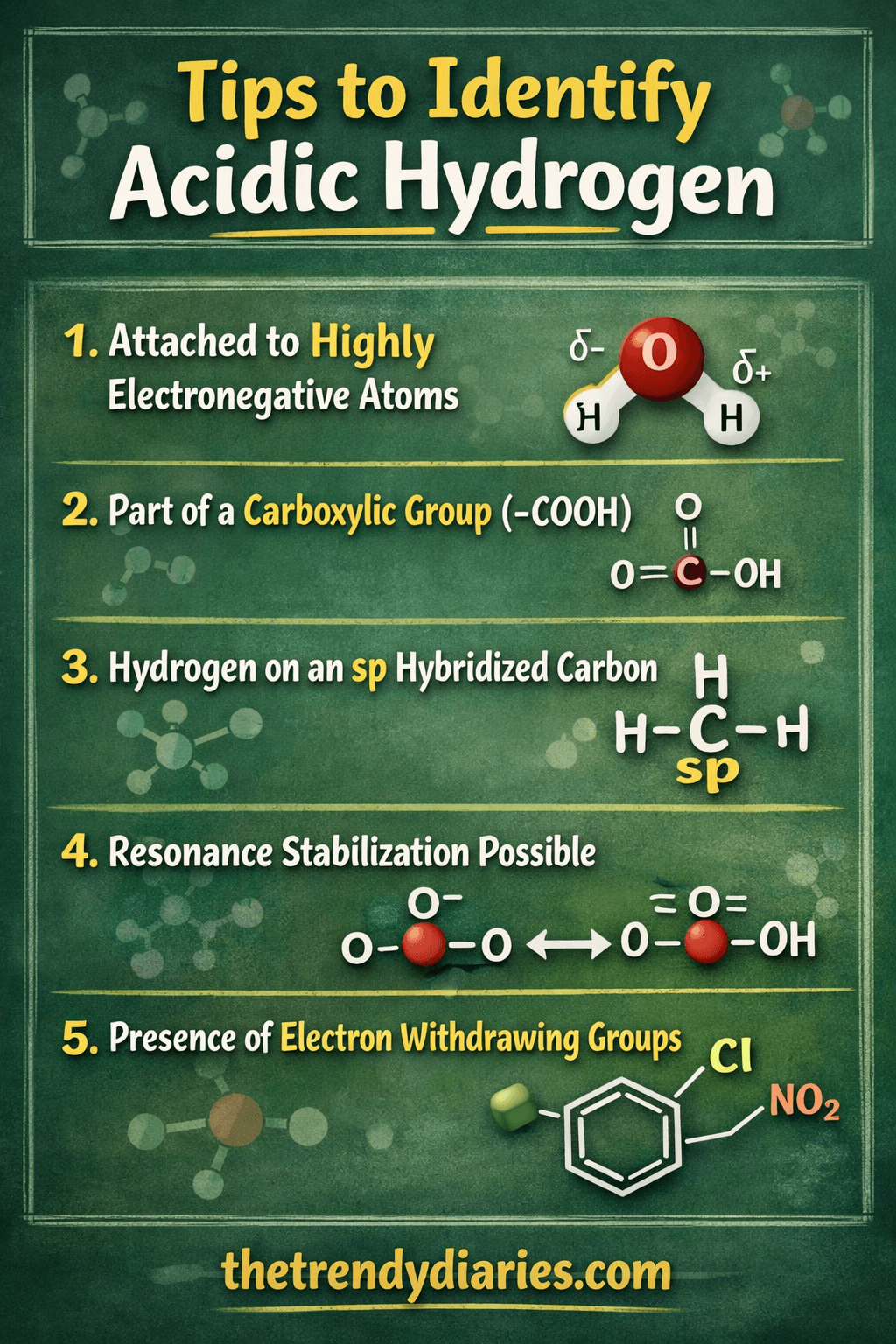
2. General Rules to Identify Acidic Hydrogen
(a) Connected Electronegative Atoms
- Hydrogens that bond to nitrogen, oxygen, or halogens are often acidic because the electronegative atom holds the negative charge upon the donation of a proton.
- Examples:
- Alcohols (-OH): H in -OH is a little acidic.
- Carboxylic acid (-COOH): H in -COOH is extremely acidic.
- Phenols the H is attached to an aromatic ring. is acidic.
b) Alpha Hydrogens
- Hydrogens bound to carbon atoms adjacent to the carbonyl group (C=O) are acidic.
- A conjugate base (enolate Ion) is stabilized by resonance, which allows for deprotonation to be made easier.
- Example A: In Acetone (CH3-CO-CH3) The hydrogens that are on the alpha carbon are acidic.
C) Hydrogens in bonds with strongly polarized bonds
- Hydrogen bonds to atoms that have an electronegativity that is high in the bond is acidic.
- Example The H component of HF (hydrogen fluoride) or H in HCl (hydrogen chloride) is acidic in water solution.
3. Methods to Test Acidic Hydrogen
a) Utilizing the pKa Values
- pKa is a measure of the potential of a substance to give the proton.
- Lower pKa indicates stronger acidity.
- Example:
- Carboxylic acid (pKa 4-5) is a highly acidic hydrogen
- Alcohol (pKa 16-18) Hydrogen that is weakly acidic
B) Action with bases
- The acidic hydrogens in the water react solid bases Like NaOH like NaOH NaH:
- Example: CH3COOH + NaOH – CH3COONa + H2O
- In this case, the hydrogen from COOH is transferred to OH-, which forms water.
- An observation: The formation of an gas evolution or salt could be a sign of hydrogen in acidic form.
c) Utilization of Indicators
- Acidic hydrogens may release protons when in an aqueous solution Change the Color of acid-base indicators :
- Litmus is red in acidic solutions and blue in basic solutions
- Phenolphthalein colorless when in an acidic medium. It’s pink when basic medium
(d) Spectroscopic Methods
- NMR Spectroscopy Acidic hydrogens show up in the downfield (higher D values) due to deshielding.
- Infrared Spectroscopy Stretching vibrations of O-H or N suggest hydrogens that could be acidic.
4. Practical Tips for Identification
| Feature | Acidic Hydrogen Present | Non-Acidic Hydrogen |
| Bonded Atom | O N, Cl close to C=O | C-H in alkanes |
| Reactivity to Base | Reacts quickly | No response |
| pKa Value | The low (strong acid) | The High (weak acid) |
| Spectroscopic Observation | Downfield in NMR O-H stretch in IR | A typical C-H shift, with no O-H stretch |
- Rule of thumb: Hydrogens that are attached to electronegative carbon atoms, and activated carbons are typically acidic.
Final Thoughts
The identification of acidic hydrogen is crucial for understanding chemical reactivity chemical reactions, as well as molecular interactions. Through analyzing the bond type electronegativity, alpha position and resonance stabilization scientists can identify which hydrogens within a molecule are acidic. Tests in the laboratory, such as the determination of pKa, base reactions indicators, as well as spectroscopic techniques, provide concrete proof. Understanding these concepts is vital for students, chemists and researchers who work on organic and inorganic and biochemical chemical compounds.